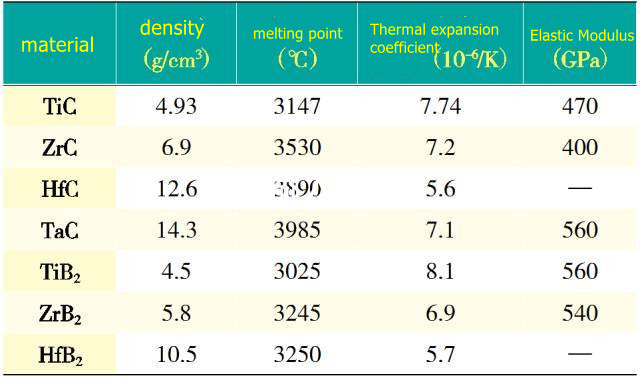
Ultra-high temperature borides mainly include hafnium boride (HfB2), zirconium boride (ZrB2), tantalum boride (TaB2) and titanium boride (TiB2), etc. Currently, zirconium boride (ZrB2) and hafnium boride (HfB2) research is the most concentrated. Boride ultra-high temperature ceramics (UHTCs) are composed of strong covalent bonds and have the characteristics of high melting point, high hardness, high strength, low evaporation rate, high thermal conductivity and electrical conductivity. It has the disadvantage of being difficult to sinter and densify. In order to improve its sintering performance and increase the density, it can be solved by increasing the surface energy of the reactants, reducing the grain boundary energy of the product, increasing the bulk diffusivity of the material, accelerating the transfer rate of the material, and improving the mass transfer kinetics.